Pfizer has announced positive top-line data from a pivotal Phase III study of abrocitinib, an investigational oral Janus kinase 1 (JAK1) inhibitor.
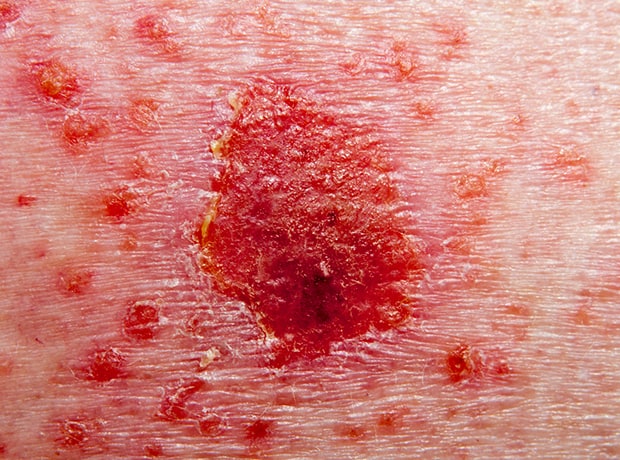
The treatment, which is being evaluated in patients aged 12 and older with moderate to severe atopic dermatitis (AD), demonstrated improvement in skin clearance and itch relief compared to placebo.
Safety data from the trial also showed that both evaluated doses of the drug – 200mg and 100mg – were well tolerated and were consistent with a companion study (JADE MONO-2), the firm noted.
There is a “critical need” for additional treatment options for patients living with moderate to severe atopic dermatitis, said Michael Corbo, chief development officer of inflammation & immunology at Pfizer.
He explained that the company is “pleased by these findings, which together with the recently reported positive top-line results from our second Phase III trial, encourage us that, if approved, abrocitinib may provide the first oral, once-daily treatment option for these patients.”
Abrocitinib received Breakthrough Therapy designation from the US Food and Drug Administration (FDA) for the treatment of patients with moderate to severe AD in February 2018, meaning that the FDA looks to expedite the development and review of the drug.
AD is a chronic skin disease characterised by inflammation of the skin and skin barrier defects. It is one of the most common, chronic, relapsing childhood dermatoses, affecting up to 10% of adults and up to 20% of children worldwide.