Eli Lilly has announced that Taltz (ixekizumab) has met the primary and all major secondary endpoints up to week 12 in the Phase IV IXORA-R study.
The company’s moderate to severe plaque psoriasis drug was evaluated against Tremfya (guselkumab) in people living with the skin disorder, and demonstrated superiority in the proportion of patients achieving complete skin clearance.
The IXORA-R trial is the first completed head-to-head trial between an IL-17A inhibitor and an IL-23/p19 inhibitor using the Psoriasis Area Severity Index (PASI) 100 score as the primary endpoint. The resulting data showed that Taltz met all major secondary endpoints up to week 12, which include superiority over Tremfya in the proportion of patients achieving PASI 75 at Week two, PASI 90 at Weeks four and eight, PASI 100 at Weeks four and eight.
Completely clear skin and rapid relief of symptoms are “possible for many people living with moderate to severe plaque psoriasis, and should be two topics dermatologists discuss with their patients,” said Andrew Blauvelt, dermatologist and president of Oregon Medical Research Center in Portland. “Head-to-head data like these are important and will help inform individual treatment goal discussions between healthcare providers and their patients.”
A total of 1,027 patients were enrolled in the study, which reported a consistent safety profile of Taltz with previous studies.
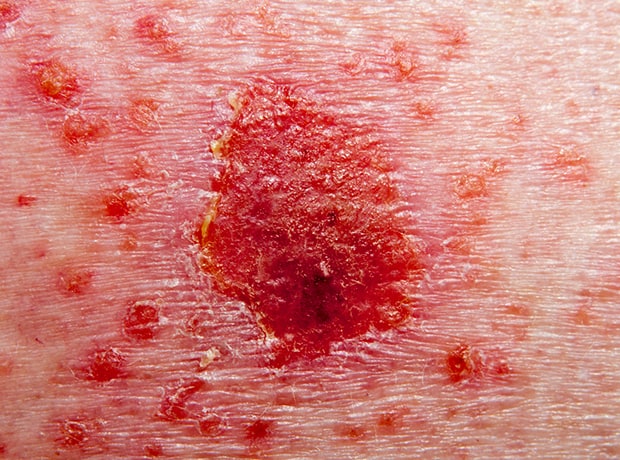
Plaque psoriasis is the most common form of psoriasis, with 80-90% of people with psoriasis experiencing plaque psoriasis. It typically appears as raised, red patches covered with a silvery white buildup of dead skin cells or scale.